Clinical Trials Rental & Maintenance Program
Rent specular microscopes & other Konan devices for your Clinical Trials.
Equipment standardization, pre-study device calibration, training specific to your trial, full documentation, and superior service and support throughout the entire study term. It’s easy.
People | vision | inspiration | innovation | dedication
Best Practices, Superior Clinical Tools, Industry Leading Service
Virtually all major ophthalmic medical device and pharmaceutical companies, and many startups in the ophthalmic space, take advantage of Konan Medical’s Clinical Trials Rental and Maintenance Program. Others choose to purchase equipment outright and contract with Konan for an ongoing extended priority support and maintenance agreement to provide assurance throughout the study period.
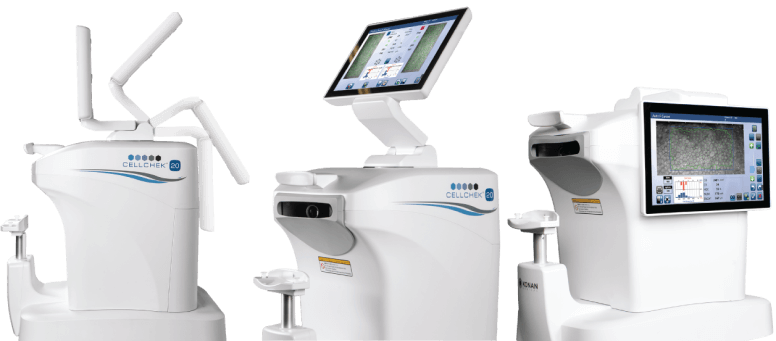
Virtually every key manufacturer of ophthalmic implantable devices and pharmaceutical agents has independently assessed options for robust measures of ECD as a primary safety endpoint for FDA clinical trials, and most have chosen Konan to capture these critical data points.

Primary Safety Endpoints for Clinical Trials
We work hard to tailor a program that helps you meet these important recommendations in a comprehensive, cost-effective, and low stress manner.
Leading Clinical Trials Program
Whatever procurement strategy you ultimately select for your study, our goal is to partner with you to find the solution that works best for you for managing and standardizing across sites in the USA or globally:
Equipment model: |
All Konan non-contact specular microscopes are FDA 510(k) cleared. |
Software version: |
Standardized across multiple sites and compatible with software at reading center. |
Technician training: |
Konan works with several reading centers to develop detailed site instructions to standardize our technical training inclusive of documentation. |
Device calibration: |
On-site and delivery of appropriate documentation. |
KonanCare highest priority support: |
Systems are provided with permission-only remote support to troubleshoot and fix imaging and imaging issues, often while the study subject is still available on site. Service is prioritized to minimize downtime. |
